Edta 4 Is Best Described as a
The species HY3 H2Y2 and so on predominate at lower pH. Chemistry questions and answers.
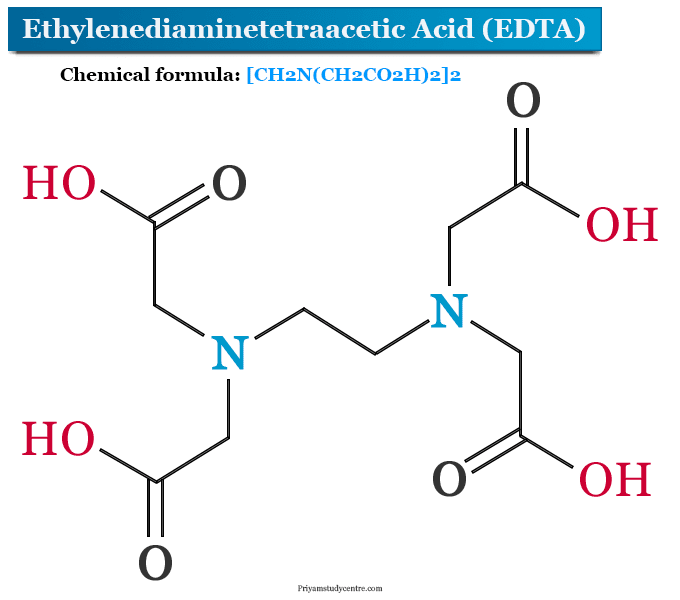
Ethylenediaminetetraacetic Acid Edta Formula Uses
Median response time is 34 minutes for paid subscribers and may be longer for promotional offers.

. EDTA4- is ethylenediamine tetraacetate ionthe total number of N-Co-O bond angles in CoEDTA-1Complex ionA4B6C8D2EDTA ion is hexa dentate. 30 in a box with a FREE StopReabsorb bowel cleanse an extra 975 value Now comes with a FREE StopReabsorb bowel cleanse that greatly supports the benefitial. When this ion bonds to a metal atom the two N atoms and four of the O atoms are used.
EDTA binds minerals and it is especially attracted to those with a 2 or 3 charge. Many of the resulting coordination compounds adopt octahedral geometry. From the definition αY4 Y4EDTA we can express the concentration of Y4.
For this purpose ironII sulfate is dissolved in 300 mL water with the addition of 4 mL sulfuric acidEDTA is dissolved in 300 mL water and 280 mL KOHThe ferrous sulfate and EDTA solutions are pooled stirred and actively aerated in the. EDTA is described as a hexadentate chelating agent hexadentate referring to 6 teeth a hint at the etymology of the word chelating. EDTA 4 usually binds to a metal cation through its two amines and four carboxylates.
The true statement amongst the following. When cells reach 7080 confluence dissociate the cells with EDTA as described in Steps 3 and 4. Edta is a hexadentate ligand because of its competence to denote six pair of lonely electrons due to the formation of a covalent bond.
Circle one 1 monodentate 5 octadentate 2 bidentate 3 tetradentate 4 hexadentate 6. The most widely used synthesis is the alkaline cyanomethylation of ethylenediamine by means of sodium cyanide and formaldehyde. Ethylenediaminetetraacetic acid EDTA is a chelating agent can bind to metals via four carboxylate and two amine groups.
The formation of FeIII-EDTA FeY can be described as. Response times may vary by subject and question complexity. It is produced as several salts notably disodium EDTA and calcium disodium EDTA.
EDTA4-forms very stable complexes with most of the transition metals. It belongs to a class of molecules called chelators from the Greek word chelé meaning claw that bind chelate metals 1. In this complex a single EDTA 4- ion forms 6 bonds to the Fe atom ie 2 Fe-N bonds and 4 Fe-O bonds.
Ethylenediamine tetraacetic acid EDTA is a polyprotic acid containing four carboxylic acid groups and two amine groups with lone-pair electrons that chelate calcium and several other metal ions. The triangle formed by the lines x y - 4 0 3x y 4 x 3y 4 is. If its area is 2 then the value of b is.
The best available evidence does not support the therapeutic use of EDTA chelation therapy in the treatment of cardiovascular disease. As seen in the fractional composition diagram most EDTA is not Y4 below pH 1037. This hexadentate ligand forms very stable complexes usually octahedral structures with most of the transition metals.
The EDTA is best described as aligand. The following reaction describes the synthesis of EDTA. The formation constant Kf MYn 4MnY4 describes the reaction between Y4 and a metal ion.
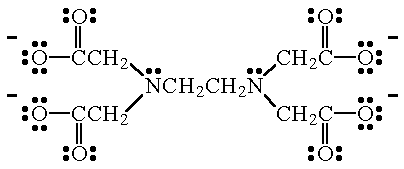
Although not considered to be a highly invasive or harmful therapy it is possible that the use of EDTA chelation therapy in lieu of proven therapy may result in causing indirect harm to the patient. An example is ethylenediamminetetraacetate ion EDTA4- the Lewis structure of which is shown below. PBS buffer contains 137 mM sodium chloride 7 mM sodium phosphate dibasic 3 mM potassium chloride 14 mM potassium phosphate monobasic 4 mM EDTA pH 74Biological buffers are inert and have short-term effects on the system under investigation both of which are distinct advantage over inorganic substances and weak organic acids that were used in the past.
The coordination number of 6 results in an octahedral structure. It is a polyamino carboxylic acid and a colorless water-soluble solid which is widely used to dissolve lime scale. A EDTA called as ethylenediaminetetraacetic acid consisting of 2 amino groups and four carboxyl groups called as Lewis bases.
H 2 NCH 2 CH 2 NH 2 4 CH 2 O 4 NaCN 4 H 2 O NaOOC CH 2 2 NCH 2 CH 2 N NaOOC CH2 2 4 NH 3. Experts are waiting 247 to provide step-by-step solutions in as fast as 30 minutes. Calcium is necessary for a wide range of enzyme reactions of the coagulation cascade and its removal irreversibly prevents blood clotting within the collection tube.
H 2 O KHSO 4 55 H 2 O. In coordination chemistry EDTA 4 is a member of the aminopolycarboxylic acid family of ligands. EDTA is a chemical that binds and holds on to chelates minerals and metals such as chromium iron lead mercury copper aluminum nickel zinc calcium cobalt manganese and magnesium.
The donor atoms in EDTA 4- are the two N atoms and the four negatively charged O atoms. Cesco Solutions TETRASODIUM EDTA - Water Softener - Tetra Sodium EDTA Powder - EDTA Chelating Agent - Sequesters Metal Ions - Reduces Limescale - Resealable Easy Pour Package 5 lbs 43 out of 5 stars. EDTA was first discovered in 1935 by Ferdinand Munz.
When they are bound they cant have any effects on the body and they are removed from the body. Some ligands can bond to a metal atom using more than two pairs of electrons. The term ligand is used to describe the number of ligand atoms used to bind a metal ion to form a metal-ligand complex.
The triangle formed by the tangent to the curve fx x 2 bx - b at the point 1 1 and the co-ordinate axes lies in the first quadrant. When they are bound they cant have any effects on the body and they are removed from the body. EDTA 20 CaNa2EDTA 2000mg Calcium Disodium EDTA.
The EDTA4 reaction with the Cu2 an example of a redox reaction. It forms coordinate bonds with a central metal atom such as iron at the right pH. Based on this description which best describes EDTA as a ligand when it binds with the Ca2 ion.
EDTA is a chemical that binds and holds on to chelates minerals and metals such as chromium iron lead mercury copper aluminum nickel zinc calcium cobalt manganese and magnesium. The tendency to form such chelates is. FeSO 4 7H 2 O K 2 H 2 Y 14 O 2 KFeYH 2 O.
How Does It Work. Based on this description which best describes EDTA as a ligand when it binds with the Ca2 ion. When they are bound they cant have any effects on the body and they are removed from the body.
Up to 12 cash back Free Shipping Worldwide US over 100 and International retail orders over 300 Detoxify or Die Sherry A. Meanwhile prethaw 200 μl of Matrigel on ice and dilute it. EDTA is a chemical that binds and holds on to chelates minerals and metals such as chromium iron lead mercury copper aluminum nickel zinc calcium cobalt manganese and magnesium.
EDTA Ethylenediaminetetraacetic acid is a transparent water-soluble compound first synthesized in 1935.

Comments
Post a Comment